At the phenotypic level, evolutionary biologists have long observed that tradeoffs place a major constraint on organisms, manifesting as negative correlations between traits or between fitness values in different environments. Thus, a population adapted to one environment may be unable to quickly adapt to a second environment. Such tradeoffs are thought to explain various phenomena such as ageing and the existence of both specialists and generalists. While a substantial body of work demonstrates negative trait correlations across species, we have relatively poor understanding of tradeoffs during the early stages of divergence, as different populations adapt to distinct environments.
To address this, we conducted a large analysis of intra-specific trait variation across 20 wild-collected flour beetle populations (Tribolium castaneum). Surprisingly, we did not find negative correlations between various fitness-related traits, or between reproductive fitness across alternative food sources (Agashe et al, in preparation). The only exception was a negative correlation between immune function and interspecific competitive ability. These results are unexpected because the populations show significant divergence for all measured traits, and yet do not exhibit major tradeoffs. Thus, fitness tradeoffs do not seem to constrain trait evolution during the early stages of population divergence.
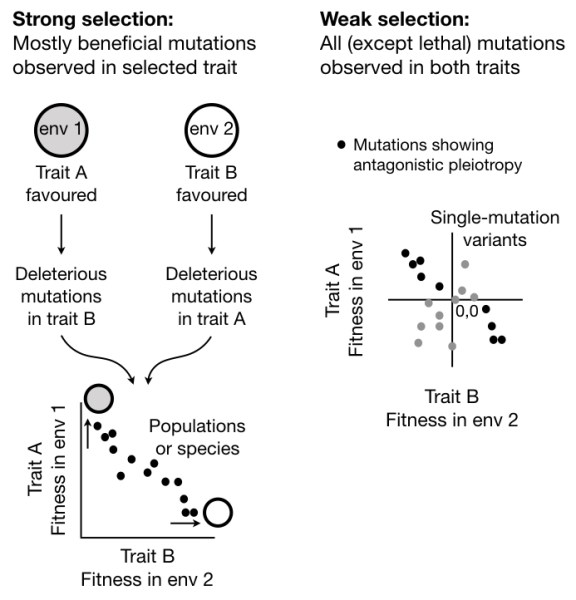
At the broader phenotypic level, tradeoffs are an undeniable outcome of resource allocation. One mechanism causing tradeoffs is antagonistic pleiotropy; i.e. when a single mutation has opposite, pleiotropic effects on two distinct phenotypes. However, we have limited understanding of the incidence and magnitude of antagonistic pleiotropy for single mutations. Previous analyses focused on microbial populations evolving under strong selection, a scenario in which a large fraction of mutations – deleterious, neutral, and beneficial mutations with small fitness effects – are poorly represented. Hence, our understanding of the fitness effects of mutations is biased towards large-effect beneficial mutations. To address this gap, we obtained unbiased estimates of pleiotropy for nearly all single mutations occurring under weak selection (under mutation accumulation) in laboratory-evolved E. coli populations (Sane, Miranda and Agashe 2018). We find that although synergistic pleiotropy is fairly common, antagonistic pleiotropy is very rare and varies substantially across 11 different carbon sources. Notably, the frequency of antagonistic pleiotropy is much lower than null expectations derived from randomly sampling the independent distributions of fitness effects for each resource. Thus, in contrast to current assumptions, most mutations are unlikely to be antagonistically pleiotropic across environments.
